Safe, Efficient, High Quality, and Compliant
Clinical phase I, II, III pilot and commercial production of drug substances (including ADCs, RDCs, PDCs, AOCs, and other drug conjugates)

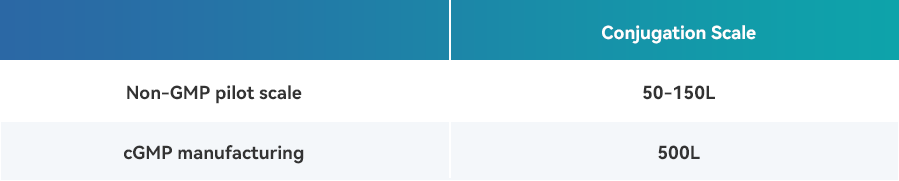
· Facilities equipped with bioreactors scaling up to 500L adapt to various conjugation processes
· Disposable materials and consumables are product specific, preventing cross-contamination
· High-potency materials up to OEB 5 and photosensitive drug conjugates could be handled in GMP facilities
· Efficient delivery: Capable of completing project production in 3-10 days on average
· Over 10 years of production experience in conjugation, purification, drug substance preparation, and filling.