


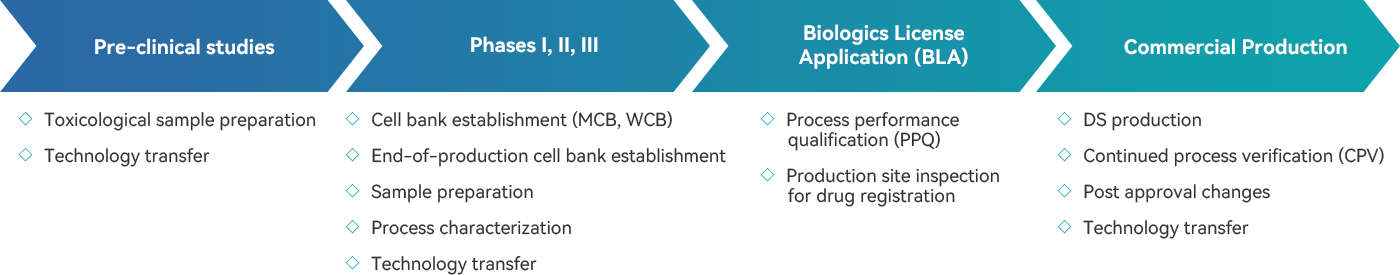
· cGMP-compliance manufacturing capacity at scales of 50L, 200L, 500L, and 2000L to meet our clients’ production needs for toxicological studies, clinical trials, and commercialization
· Continuous expansion of commercial manufacturing capacity in Fengxian, Shanghai
· Platformized materials to effectively reduce the risk of RA and compliance while maintaining product quality
· The professional and experienced manufacturing, science & technology (MSAT) team to ensure technology and production and success
· Provides rapid, comprehensive, and effective technology transfer solutions for clinical sample preparation and commercial production
· Over 10 years of clinical and commercial production experience
· Successfully completed GMP batch production for multiple projects