


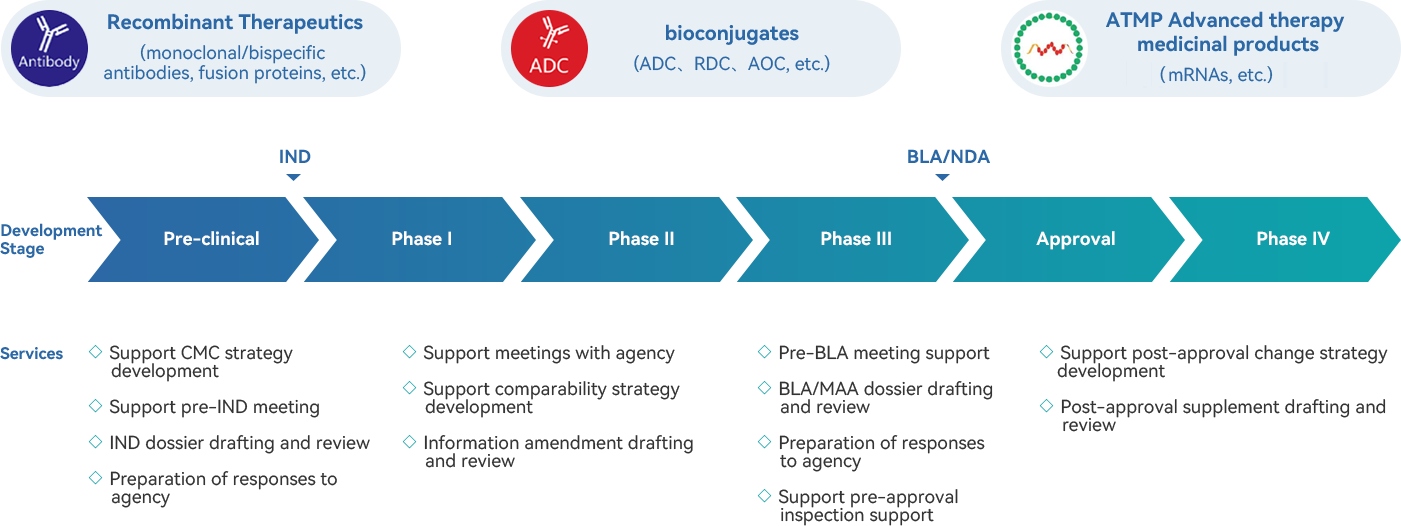
· Comprehensive CMC dossier template, regulation/guideline and CMC knowledge sharing platform. Continually monitor and interpret laws/ regulations/ guidelines to ensure product compliance throughout the lifecycle
· Core team members came from health authority, MNC or leading CDMO, with over 25yr biologics registration and regulation experience
· Core team members frequently share expertise at industry conferences, actively participate in shaping the regulatory environment shaping, and have established strong connections with health authorities and industry associations
· Registration experience for multiple kinds of biologics, including eukaryotic and prokaryotic expression products, conjugation products, mRNA, etc.
· Extensive experience in dossier preparation for biologics IND, BLA, and PAC application